
OMAX MD82ES10 Digital LED Compound Microscope
About OMAX
OMAX Corporation is a well-known manufacturer of high-precision abrasive waterjet systems. Founded in 1993 and headquartered in Kent, Washington, USA, OMAX has established itself as a leading provider of waterjet cutting technology. Waterjet cutting is a versatile and precise method of cutting a wide range of materials using a high-pressure jet of water mixed with abrasive particles.
Instructions
Caution
- Open the carton carefully with a knife or paper cutter. Find the “UP” sign and place the Styrofoam container on the side that makes the arrow upward. If the “UP” sign is missing, please open the Styrofoam container gently to prevent any accessory, i.e. objectives or eyepieces, from dropping and being damaged.
- Do not discard the molded Styrofoam container; the container should be retained should the microscope ever require reshipment.
- Keep the instrument out of direct sunlight, high temperature or humidity, and dusty environments. Ensure the microscope is located on a smooth, level, and firm surface.
- If any specimen solutions or other liquids splash onto the stage, objective, or any other component, disconnect the power cord immediately and wipe up the spillage. Otherwise, the instrument may be damaged.
- All electrical connectors (power cord) should be inserted into an electrical surge suppressor to prevent damage due to voltage fluctuations.
- Confirm that the input voltage indicated on your microscope corresponds to your line voltage. The use of a different input voltage other than that indicated will cause severe damage to the microscope.
- Note: please read the instructions for the operation of the camera in manual 3.9 below and the CD in the package before you start to use it.
Care and Maintenance
- Do not attempt to disassemble any component including eyepieces, objectives, or focusing assembly.
- Keep the instrument clean; remove dirt and debris regularly. Accumulated dirt on metal surfaces should be cleaned with a damp cloth. More persistent dirt should be removed using a mild soap solution. Do not use organic solvents for cleansing.
- The outer surface of the optics should be inspected and cleaned periodically using an air stream from an air bulb. If dirt remains on the optical surface, use a soft cloth or cotton swab dampened with a lens cleaning solution (available at camera stores). All optical lenses should be swabbed using a circular motion. A small amount of absorbent cotton wound on the end of a tapered stick makes a useful tool for cleaning recessed optical surfaces. Avoid using an excessive amount of solvents as this may cause problems with optical coatings or cemented optics or the flowing solvent may pick up grease making cleaning more difficult. Oil immersion objectives should be cleaned immediately after use by removing the oil with lens tissue or a clean, soft cloth.
- Observe the specimen with the 4X, 10X, and 40X objectives in order, then observe the specimen with the 100X objective. Apply the immersion oil on the slide cover with the 100X objective. Do not let the immersion oil to contact with the dry objectives lens (especially the 40X). Clean the dry objective lens using the lens cleaning paper if the immersion oil is on the dry objective lens. Clean the 100X objective lens first using the lens cleaning paper after observing the specimen with the 100X objective, then clean the specimen. More persistent dirt should be removed using a little bit of alcohol. Do not use organic solvents for cleansing.
- Store the instrument in a cool, dry environment. Cover the microscope with the dust cover when not in use.
Components Illustration
- Eyepiece
- Diopter Adjusting Ring
- Eyepiece Tube
- Nosepiece
- Objective
- Slide Holder
- Mechanical Stage
- Light Collector
- Microscope Base
- Viewing Head w Built-in Camera
- Microscope Body
- Focusing Rack Stop Screw
- USB Cable
- Coarse Focus Knob
- Fine Focus Knob
- X-Y Stage Moving Knobs
- Power Switch
- Brightness Intensity Dial
- Abbe Condenser
- Condenser Lock Thumb Screw
- Condenser Control Knob
- Condenser Holder
- Color Filter Holder
- Iris Diaphragm Lever
Installation
Installation of the eyepieces
- Remove the protective caps from the eyepiece tubes.
- Insert the eyepieces into the eyepiece tubes.
Installation of the glass filter
- Swing out the color filter holder under the condenser.
- Place the filter into the holder as shown in Fig. 1, and swing the holder in.
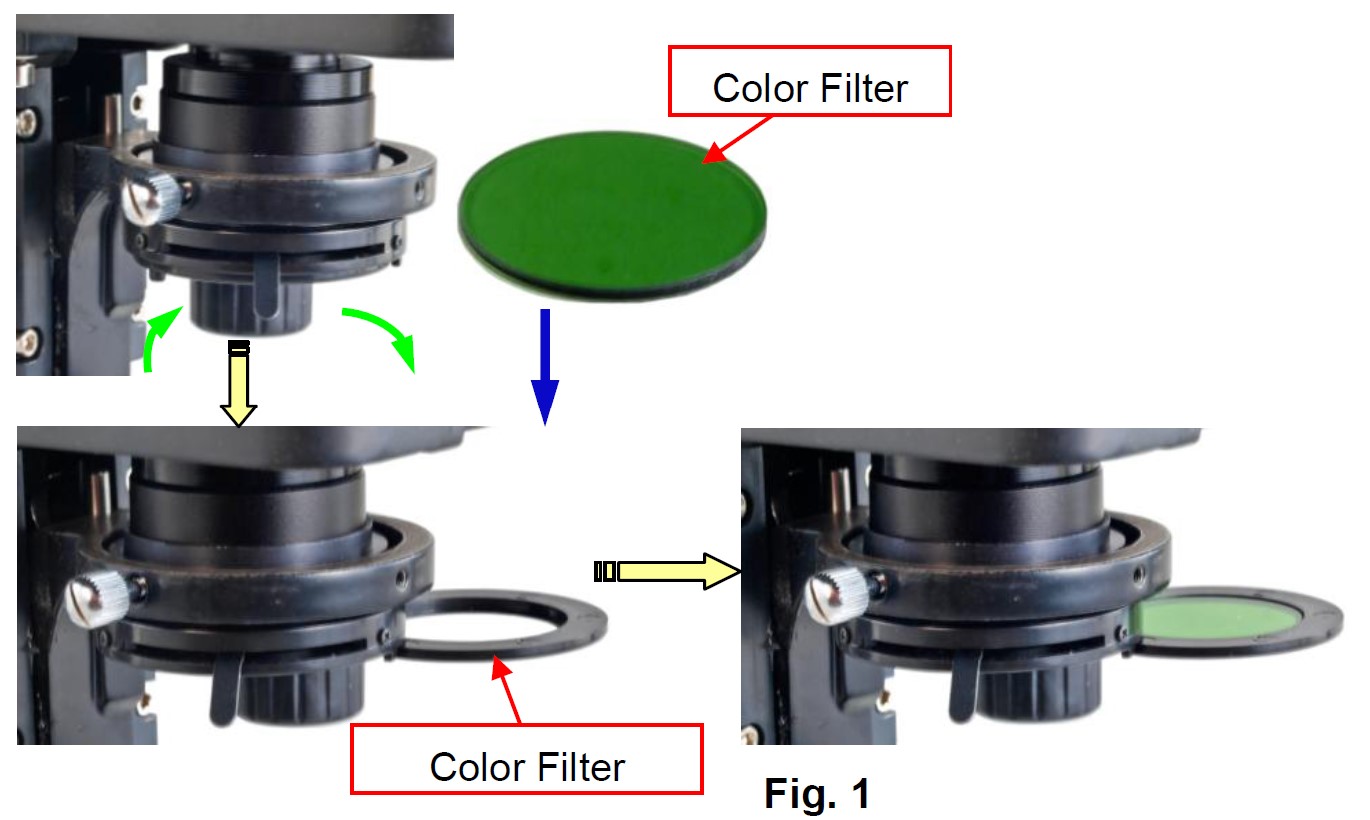
Operation
Adjusting illumination
- Plug the power cord into the power socket on the microscope and connect it to the power outlet.
- Turn on the power switch.
- Rotate the brightness intensity dial to increase or decrease the brightness.
Caution: A diffusion filter is attached beneath the condenser to get uniform light and protect your eyes from strong LED light when a low-power objective applies. The diffusion filter can be swung out to make the view field brighter when observing with a high-power objective, such as a 100X objective.
Placing specimen
- Place the slide on the mechanical stage.
- Use the slide holder to gently secure the slide.
- Turn the X and Y stage moving knobs to position the specimen in the center of the viewing field.
Caution: Be sure not to allow an objective to touch a specimen slide when changing objectives.
Adjusting interpupillary distance
- While observing with both eyes, hold the left and right eyepiece tubes and slide the tubes in and out.
- The interpupillary distance is correct when the left and right fields of view converge completely into one image.
Adjusting eyepiece diopter
- Rotate the 10X objective into position.
- Rotate the diopter ring on the right eyepiece tube until its numerical value is the same as your interpupillary distance, for example, 70 in the right figure (See Fig. 2).

- Close your left eye and bring the specimen into focus following the focusing procedures in 3.5.
- Close your right eye and bring the same specimen into clear sharp focus by adjusting the diopter ring on the left eyepiece tube only. Do not use focus knobs at this step.
- Since both sides are adjustable, you may also do the above in the opposite way, in other words, left eye first and right eye second.
Focusing
- With the 10X objective in position, raise the mechanical stage using the coarse focus knob until the specimen is close to the objective
- Turn the coarse focus knob until the specimen is in focus. Use the fine focus knob to obtain a sharp image. You may now switch to another magnification objective.
- To get a good focused image, you may need to combine the focus knob adjustment and interpupillary distance adjustment, along with the eyepiece
Focusing Rack Stop diopter adjustment stated in 3.3 and 3.4.
Tips:
- To prevent your specimen slide from making contact with an objective, turn the 100X objective in position and adjust the focusing rack stop screw (Fig. 3) so that the 100X objective will not contact the specimen while the stage is adjusted to its highest position.

- Give the stage a tiny extra moving space to ensure the objective can be focused every time.
Applying the immersion oil
- Rotate the objective nosepiece to seat the observing position between the 40X and 100X objectives as shown in Fig. 4 (a).
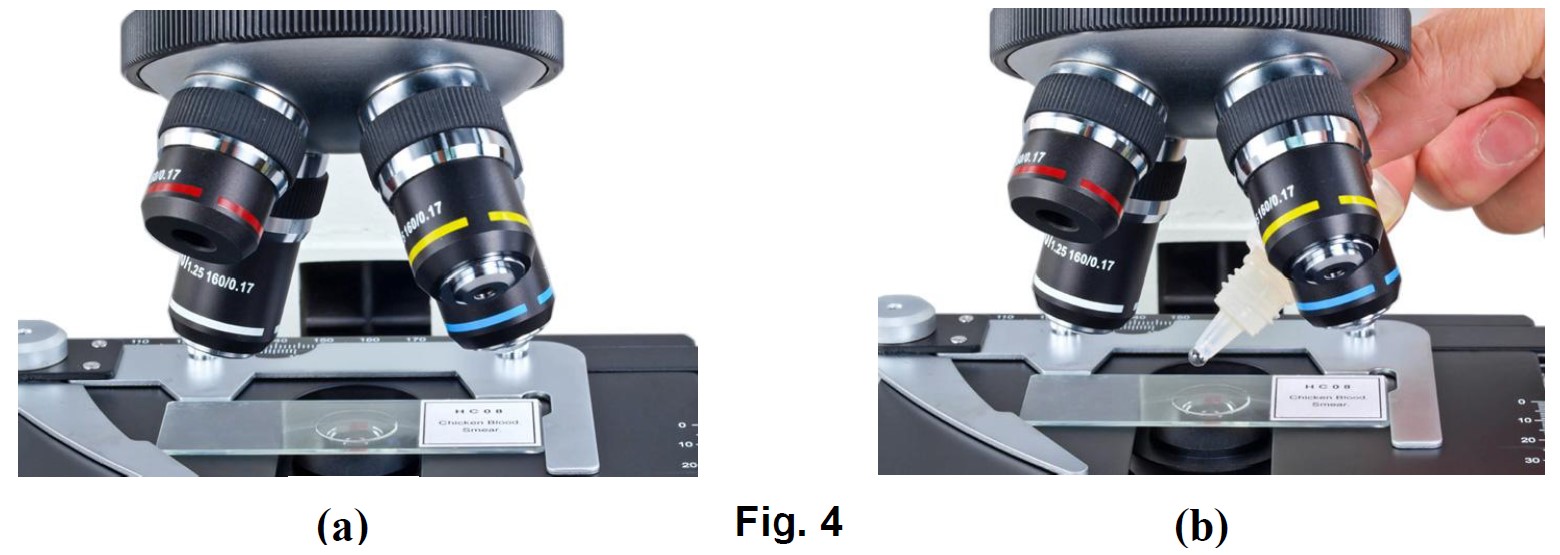
- Place a drop of immersion oil on the slide cover as shown in Fig. 4 (b).
- Rotate the objective nosepiece to seat the 100X objective to the observing position until you hear a “click” sound.
- After observing the specimen, use the lens cleaning paper to clean the 100X objective lens gently and the specimen in time.
- If it is hard to clean, you need a little bit of alcohol to clean the 100X objective lens and the specimen.
Caution (Important):
- When you use the 100X objective to observe the specimen, you have to finish observing the specimen with the 4X, 10X, and 40X objectives.
- When you use the 100X objective to observe the specimen, you have to apply the immersion oil on the top of the slide cover.
- When you apply the immersion oil with the 100X objective, do not let the immersion oil to contact with the dry objective lenses (especially the 40X). If the immersion oil is on the dry objectives lens, please use the lens cleaning paper to clean the objectives lens in time. The oil will damage the dry objective lenses.
- After observing the specimen with the 100X objective, clean the 100X objective lens first.
Adjusting condenser
- Turn the condenser focus knob to raise or lower the condenser.
- Raise the condenser when using high-power objectives and lower it when using low-power objectives.
Note:
- The centering of the condenser and the light axis of the objective are factory-adjusted. Do not attempt re-adjustment.
- The highest position of the condenser has been factory-adjusted. Do not attempt re-adjustment.
Adjusting the iris aperture diaphragm
Swing the iris diaphragm lever (Fig. 5) left or right to adjust the aperture size.
Note: The iris diaphragm is designed to adjust the aperture size, not to adjust the brightness although the brightness will be changed when it’s adjusted. When the aperture is adjusted to a smaller size, the contrast will be increased and the depth of field will be increased as well. Turn up the intensity of the light if the image is too dim.
Photo/video observing, capturing, and recording
- Bring the microscope into focus by following the procedures in 3.5.
- Insert the USB cable into the USB port (Fig. 6) on the back of the viewing head, and the other end to the computer.

- Turn on the computer; install the camera following the manual on the mini CD.
- Open image observing software to examine.
- Capture images with the manual white balance for an image size of 1280X1024 or 1024X768 resolution when using auto white balance.
- You also can record live videos through the software.
Note:
- Please refer to the manual on the camera’s CD for the details of installation and operation of the camera.
- Do not capture images of 1280X1024 resolution in auto white balance mode. The captured images may have problems with color rendering in this mode.
Specifications
- Model: MD82ES10
- Total Magnification: 40X, 80X, 100X, 200X, 400X, 800X, 1000X, 2000X
- Viewing Head : Binocular, inclined 45º, 360º swiveling with Built-in camera
- Interpupillary Distance: Sliding adjustment, 2-3/16” ~ 2-15/16” (55mm ~ 75mm)
- Diopter Adjustment On both eyepiece tubes
- Eyepieces : 1 pair of wild field WF10X/18 1 pair of wild field WF20X
- Nosepiece: Revolving quadruple
- Objectives : Achromatic DIN 4X/0.10 160/0.17
10X/0.25 160/0.17
40X/0.65 160/0.17(spring), 100X/1.25 160/0.17(spring, oil) - Condenser: Abbe, NA=1.25, w/ iris diaphragm, and diffusion filter Rack and pinion adjustment
- Focus Mechanism: Coaxial coarse and fine focusing knobs on both sides with rack stop
- Mechanical Stage : Double layer, dimension: 4-1/2” x 4-15/16”(115 mm x 125 mm) X-Y translational range: 2-13/16” x 1-3/16” (70 mm x 30 mm)
- Illumination: Transmitted, LED, variable intensity
- Camera: Built-in USB2.0 1280 x 1024 pixel (1.3MP) Driver and Software included in the CD Compatible with Windows 2000, XP, Vista, and Windows 7 32/64-bit
- Dry Darkfield Condensers (optional): Refer to the Dry Darkfield Condensers Installation & Operation Instruction
- Power Supply: AC/DC adapter, 100V-240V,
- Dimension : 9-1/16” x 7-1/8” x 13” (23cm x 18cm x 33cm)
- Net weight: 7 lbs 5 oz (3.32 kg)
Troubleshooting Guide
Darkfield Condenser Installation and Operation Instructions
- Mounting the dry darkfield condenser
- Rotate the nosepiece and set the 4x objective in position.
- Loosen and raise the focusing rack stop screw to the highest position.
- Turn the coarse focus knob to raise the mechanical stage to the highest position without contacting the 4X objective.
- Turn the condenser control knob to raise the condenser to the highest position.
- Loosen the condenser lock thumb screw, pull down the condenser and take it off, insert the dry darkfield condenser in, and tighten the condenser lock thumb screw (See Fig. 7).

- Re-adjust the condenser height to its normal position.
- Re-adjust the mechanical stage height to its normal position.
- Lower the focusing rack stop screw back and use the bolt to lock it when needed.
Note: When raising the mechanical stage, do not make contact with the objective.
- Centering the dry darkfield condenser
- Turn the 4X objective to the light path.
- Turn the condenser focus knob to lower the condenser till a dark spot showed in the viewing field as shown in Fig. 8 (a).
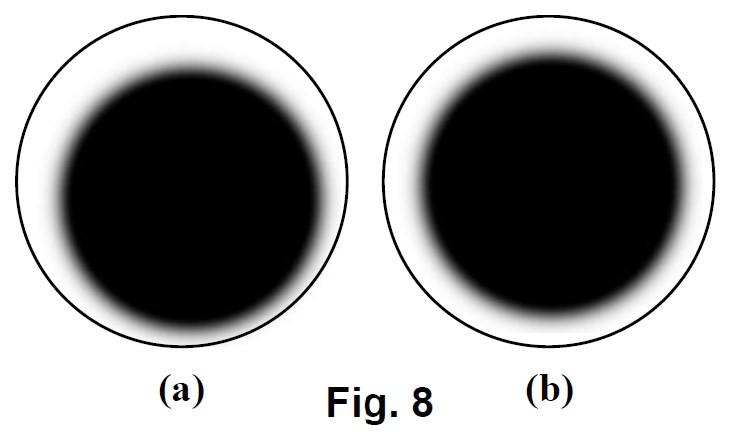
- Turn the condenser translational centering screws to move the dark spot to the center as shown in Fig. 8 (b).
- Place the slide on the stage.
- Raise the condenser all the way to the top and then lower it a little bit.
- Follow the procedures in this manual to bring the sample on a slide in focus and observe.
- Move the condenser up or down slightly to get the best darkfield viewing.
Note:
- The dry darkfield condenser is used with the dry objectives only.
- The dry darkfield condenser works with the 4X, 10X, and 40X objectives.
- The dry darkfield condenser does not work with the 100X oil immersion objective.
FAQS About OMAX MD82ES10 Digital LED Compound Microscope
How reliable are OMAX microscopes?
The OMAX is quite enjoyable to use and has high-quality aims, outstanding build and design, a ton of practical features, accessories, and customization options.
Omax is a what kind of microscope?
This simple binocular pupil microscope
What is the ideal object to observe with a compound light microscope?
Blood cells, cheek cells, parasites, bacteria, algae, tissue, and microscopic organ fragments are only a few examples.
What nation’s brand is OMAX?
Switzerland
OMAX microscopes are produced where?
China
What is the OMAX microscope warranty?
For a period of five years, microscopes
What kind of microscope is the most recent?
swirling microscope
Is light required for compound microscopes?
The sample lets through light.
How should I clean a microscope?
Use either a lens cleaning solution or 100% ethanol to get rid of greasy debris.
A compound light microscope has how many lenses?
dual lenses
For more manuals by OMAX, Visit Manualsdock


